|
|
Post by jeany on Jun 11, 2009 14:30:29 GMT -5
Effects of Bleached Pulp Mill Effluents on the Aquatic Environment www.hc-sc.gc.ca/ewh-semt/pubs/contaminants/psl1-lsp1/pulp_mill_effluents_pate_blanchie/pulp_mill_effluents_pate_blanchie_4-eng.php#a49The concentrations of compounds found in biotreated bleached effluents, excepting tetrachloroguaiacol, trichlorophenol, and chlorodehydroabietic acid, are a small fraction of the levels that are acutely lethal to fish or other aquatic life. The concentration range (ppb) of bleached pulp mill-generated compounds found in biologically-treated kraft and sulphite pulp mill effluents as well as their corresponding LC50s to aquatic organisms. Chronic Effects Unlike acute lethality, chronic toxicity normally does not result in the immediate death of an organism upon exposure to a pollutant. Chronic effects typically develop after continuous, long-term exposure to low doses of toxic material. In many instances, the effects a pollutant may exert on the individual organism, although subtle, may be important to the continuance of the species, e.g., reproduction, growth, or survival. As difficult and important as it is to identify the exact effect a compound may have on an organism, it is often even more difficult to identify the ecological significance of these often subtle responses in the population. Unfortunately, there is incomplete information on the chronic effects of individual chlorinated organic compounds discharged in bleached pulp mill effluents. Also, predicting the toxicity of a mixture of these compounds from that of the individual components, which is achievable in the case of acute toxicity, is at present a nearly hopeless task at the chronic level. Reproductive and Life-cycle Effects. Reproductive performance of aquatic organisms exposed to bleached pulp mill effluents for one or more entire life cycles should be among the most sensitive and relevant evaluations of chronic effects. Unfortunately, most researchers have conducted experiments which deal with effects only on early life-cycle stages or some limited aspect of reproduction. Biochemical and Physiological Changes. Whole bleached pulp mill effluent has been shown in field and laboratory studies to elicit biochemical and physiological changes in fish. Liver and blood parameters are most commonly examined for these biochemical and physiological disturbances. Morphology. Various degrees of skeletal deformities as well as fin and gill erosion have been reported in fish from areas near bleached pulp mill discharge. Mutagenicity. Bleached pulp mill effluents have been found to be mutagenic using standard tests. Carcinogenicity. A number of compounds found in bleached pulp mill effluents have been identified as carcinogens on the basis of standard methods of mammalian testing. Among these are chloroform, carbon tetrachloride, and safrole. Some other compounds, such as various chlorinated benzenes and phenols, epoxystearic acid and dichloromethane, have been classified as suspected carcinogens. Behaviour Modification. Limited information exists on the behavioural response of aquatic organisms to whole effluent. Unfortunately, the information that does exist cannot differentiate which fraction of bleached pulp mill effluent, such as chlorinated organic substances, BOD, dissolved oxygen, pH, turbidity or suspended fibres, is responsible for behavioural modification Species Diversity. Numerous studies have documented shifts in species dominance near pulp mills using chlorine bleaching.
Jeany |
|
|
|
Post by jeany on Jun 11, 2009 14:34:10 GMT -5
Water pollution en.wikipedia.org/wiki/Water_pollutionWater pollution is the contamination of water bodies such as lakes, rivers, oceans, and groundwater caused by human activities, which can be harmful to organisms and plants that live in these water bodies. It occurs when pollutants are discharged directly into water bodies without treating it first. Water pollution is a major problem in the global context. It has been suggested that it is the leading worldwide cause of deaths and diseases, and that it accounts for the deaths of more than 14,000 people daily In addition to the acute problems of water pollution in developing countries, industrialized countries continue to struggle with pollution problems as well. In the most recent national report on water quality in the United States, 45 percent of assessed stream miles, 47 percent of assessed lake acres, and 32 percent of assessed bay and estuarine square miles were classified as polluted. Water is typically referred to as polluted when it is impaired by anthropogenic contaminants and either does not support a human use, like serving as drinking water, and/or undergoes a marked shift in its ability to support its constituent biotic communities, such as fish. Natural phenomena such as volcanoes, algae blooms, storms, and earthquakes also cause major changes in water quality and the ecological status of water. Water pollution has many causes and characteristics. The specific contaminants leading to pollution in water include a wide spectrum of chemicals, pathogens, and physical or sensory changes such as elevated temperature and discoloration. While many of the chemicals and substances that are regulated may be naturally occurring (calcium, sodium, iron, manganese, etc.) the concentration is often the key in determining what is a natural component of water, and what is a contaminant. Oxygen-depleting substances may be natural materials, such as plant matter (e.g. leaves and grass) as well as man-made chemicals. Other natural and anthropogenic substances may cause turbidity (cloudiness) which blocks light and disrupts plant growth, and clogs the gills of some fish species. Many of the chemical substances are toxic. Pathogens can produce waterborne diseases in either human or animal hosts. Alteration of water's physical chemistry include acidity (change in pH), electrical conductivity, temperature, and eutrophication. Eutrophication is the fertilization of surface water by nutrients that were previously scarce. Chemical and other contaminants Contaminants may include organic and inorganic substances. Organic water pollutants include: * Detergents * Disinfection by-products found in chemically disinfected drinking water, such as chloroform * Food processing waste, which can include oxygen-demanding substances, fats and grease * Insecticides and herbicides, a huge range of organohalides and other chemical compounds * Petroleum hydrocarbons, including fuels (gasoline, diesel fuel, jet fuels, and fuel oil) and lubricants (motor oil), and fuel combustion byproducts, from stormwater runoff * Tree and brush debris from logging operations * Volatile organic compounds (VOCs), such as industrial solvents, from improper storage. Chlorinated solvents, which are dense non-aqueous phase liquids (DNAPLs), may fall to the bottom of reservoirs, since they don't mix well with water and are denser. * Various chemical compounds found in personal hygiene and cosmetic products Jeany |
|
|
|
Post by jeany on Jun 11, 2009 14:34:45 GMT -5
Inorganic water pollutants include:
* Acidity caused by industrial discharges (especially sulfur dioxide from power plants)
* Ammonia from food processing waste
* Chemical waste as industrial by-products
* Fertilizers containing nutrients--nitrates and phosphates--which are found in stormwater runoff from agriculture, as well as commercial and residential use
* Heavy metals from motor vehicles (via urban stormwater runoff) [13] and acid mine drainage
* Silt (sediment) in runoff from construction sites, logging, slash and burn practices or land clearing sites
Macroscopic pollution--large visible items polluting the water--may be termed "floatables" in an urban stormwater context, or marine debris when found on the open seas, and can include such items as:
* Trash (e.g. paper, plastic, or food waste) discarded by people on the ground, and that are washed by rainfall into storm drains and eventually discharged into surface waters
* Nurdles, small ubiquitous waterborne plastic pellets
* Shipwrecks, large derelict ships
Jeany
|
|
|
|
Post by kammy on Jun 11, 2009 14:35:41 GMT -5
I notice that the chollera is blue green algae...  lol... ok, like to kill like... I've got neem oil, vit c & NAC... good to go! The nice man at the health food said for me to eat cilantro, that it de-toxes us for Mercury, lead, and aluminum. |
|
|
|
Post by jeany on Jun 11, 2009 14:35:51 GMT -5
Most water pollutants are eventually carried by rivers into the oceans.
In some areas of the world the influence can be traced hundred miles from the mouth by studies using hydrology transport models. Advanced computer models such as SWMM or the DSSAM Model have been used in many locations worldwide to examine the fate of pollutants in aquatic systems. Indicator filter feeding species such as copepods have also been used to study pollutant fates in the New York Bight, for example. The highest toxin loads are not directly at the mouth of the Hudson River, but 100 kilometers south, since several days are required for incorporation into planktonic tissue. The Hudson discharge flows south along the coast due to coriolis force. Further south then are areas of oxygen depletion, caused by chemicals using up oxygen and by algae blooms, caused by excess nutrients from algal cell death and decomposition. Fish and shellfish kills have been reported, because toxins climb the food chain after small fish consume copepods, then large fish eat smaller fish, etc. Each successive step up the food chain causes a stepwise concentration of pollutants such as heavy metals (e.g. mercury) and persistent organic pollutants such as DDT. This is known as biomagnification, which is occasionally used interchangeably with bioaccumulation.
Water samples may be examined using the principles of analytical chemistry. Many published test methods are available for both organic and inorganic compounds. Frequently-used methods include pH, biochemical oxygen demand (BOD), chemical oxygen demand (COD), nutrients (nitrate and phosphorus compounds), metals (including copper, zinc, cadmium, lead and mercury), oil and grease, total petroleum hydrocarbons (TPH), and pesticides.
and on, and on, and on...
Jeany
|
|
|
|
Post by violet on Jun 11, 2009 16:28:45 GMT -5
QUOTE=Jeany Arsenic poisoning
Arsenic poisoning kills by allosteric inhibition of essential metabolic enzymes, leading to death from multi-system organ failure. It primarily inhibits enzymes that require lipoic acid as a cofactor, such as pyruvate and alpha-ketoglutarate dehydrogenase. Because of this, substrates before the dehydrogenase steps accumulate, such as pyruvate (and lactate). It particularly affects the brain, causing neurological disturbances and death. The toxicity of arsenic and its compounds is highly variable.Organic forms appear to have a lower toxicity than inorganic forms of arsenic. Research has shown that arsenites (trivalent forms) have a higher acute toxicity than arsenates (pentavalent forms) The acute minimal lethal dose of arsenic in adults is estimated to be 70 to 200 mg or 1 mg/kg/day.Most reported arsenic poisonings are not caused by elemental arsenic, but by one of arsenics compounds, especially arsenic trioxide, which is approximately 500 times more toxic than pure arsenic. Symptoms include violent stomach pains in the region of the bowels; tenderness and pressure; retching; excessive saliva production; vomiting; sense of dryness and tightness in the throat; thirst; hoarseness and difficulty of speech; the matter vomited, greenish or yellowish, sometimes streaked with blood; diarrhea; tenesmus; sometimes excoriation of the anus; urinary organs occasionally affected with violent burning pains and suppression; convulsions and cramps; clammy sweats; lividity of the extremities; countenance collapsed; eyes red and sparkling; delirium; death. Some of these symptoms may be absent where the poisoning results from inhalation, as of arseniuretted hydrogen. Symptoms of arsenic poisoning start with mild headaches and can progress to lightheadedness and usually, if untreated, will result in death. Arsenic poisoning can lead to a variety of problems, from skin cancer to keratoses of the feet. Chronic exposure to inorganic arsenic may lead to cutaneous hyperpigmentation. Industries that use inorganic arsenic and its compounds include wood preservation, glass production, nonferrous metal alloys, and electronic semiconductor manufacturing. Inorganic arsenic is also found in coke oven emissions associated with the smelter industry. Occupational exposure to arsenic may occur with copper or lead smelting and wood treatment, among workers involved in the production or application of pesticides containing organic arsenicals. Humans are exposed to arsenic through air, drinking water, and food (meat, fish, and poultry); this food is usually the largest source of arsenic. Arsenic was also found in wine if arsenic pesticides are used in the vineyard. Arsenic is well absorbed by oral and inhalation routes, widely distributed and excreted in urine; most of a single, low-level dose is excreted within a few days after consuming any form of inorganic arsenic. Remains of arsenic in nails and hair can be detected years after the exposure. Chronic arsenic poisoning results from drinking water with high levels of arsenic over a long period of time. This may occur due to arsenic contamination of groundwater. The World Health Organization recommends a limit of 0.01 mg/L (10ppb) of arsenic in drinking water. This recommendation was established based on the limit of detection of available testing equipment at the time of publication of the WHO water quality guidelines. More recent findings show that consumption of water with levels as low as 0.00017 mg/L (0.17ppb) over long periods of time can lead to arsenicosis. Jeany I think that tea (even herbal) contains arsenic, unless it's organic tea. That's what I've read, anyway. I'll bet there may still be some arsenic in organic tea, too.... |
|
|
|
Post by kammy on Jun 12, 2009 5:32:47 GMT -5
Look at this one! These are Chinese Water Samples. Chinese Water: A Picture is Worth... [Updated] : TreeHuggerChinese Water: A Picture is Worth...  I'm reading where the MSG Factory is right next to the Computer Chip manufacturer, which is right next to the paper mill, which is next to the plastic factory... etc., etc., and they are all dumping their waste water into the river. I'm wondering if some sector uses the chromium and neodymium metals right next to a cotton or paper factory? |
|
|
|
Post by kammy on Jun 12, 2009 5:38:38 GMT -5
Remember: that the sphere that was earlier identified as 'Crypto' is now being identified as cyanobacteria or in the blue green algae family in the Peridiscaceae family, the 'white specks/seeds': |
|
|
|
Post by kammy on Jun 12, 2009 5:54:15 GMT -5
ALGAE BLOOM GLERL/Sea Grant: Algae - Harmful Algal Blooms - Toxins"What are Cyanobacteria (blue-green algae)? - Blue-green algae is the common name for several different types of algae. They are actually bacteria (Cyanobacteria) which are able to photosynthesise, hence the green colour. Cyanobacteria are bacteria that grow in water and are photosynthetic (use sunlight to create food and support life). Cyanobacteria live in terrestrial, fresh, brackish, or marine water. They usually are too small to be seen, but sometimes can form visible colonies. Cyanobacteria have been found among the oldest fossils on earth and are one of the largest groups of bacteria. Cyanobacteria have been linked to human and animal illnesses around the world, including North and South America, Africa, Australia, Europe, Scandinavia, and China. Cyanobacteria are the most common, but not the only, group of algae to form HABs. What causes an algal bloom? - There is no single factor which causes an algal bloom. A combination of optimum factors such as the presence of good nutrients, warm temperatures and lots of light all encourage the natural increase in numbers of blue-green algae in our waterways. Nature mostly takes care of the temperature and light, but the increased presence of nutrients such as phosphorous is largely due to poor farming practices such as high use of fertilizers and presence of livestock near water supplies, as well as effluent and run-off from towns and cities near waterways. The ponding of water and reducing river flow rates tends to improve the light and sometimes the nutrient environment for algal growth making water turbulance a major factor in bloom develpment. Pesticides and other chemicals may affect the natural grazers which would otherwise control algal growth and their presence increases the risk of blooms." |
|
|
|
Post by kammy on Jun 12, 2009 6:43:34 GMT -5
|
|
|
|
Post by kammy on Jun 12, 2009 10:13:24 GMT -5
I've made an attempt to tell 7 doctors about the potentially harmful toxins in a certain brand of toilet paper, a brand of ear buds, and suspected other products, this is documented in my medical records. Relaying this information to my family and doctors has caused them to become concerned about me and progressed my 'DOP' status to higher grounds! Not fun!
This appears to be a losing battle to approach it the way it's supposed to be done. I think I need to print some information and mail it anonymously to the local Health Departments in 2 counties, at least, and possibly to the CDC?
|
|
|
|
Post by jeany on Jun 12, 2009 11:03:50 GMT -5
Family of Cyanobacteria 1. Acaryochloris marina 2. Lyngbya majuscula 3. Aphanizomenon 4. Anabaena 5. Chroococcidiopsis Acaryochloris marina. Acaryochloris marina was first isolated as an epiphyte of algae. Strains of A. marina been isolated from a variety of habitats and locations, usually associated with algae but also as free-living organisms. This cyanobacterium produces an atypical photosynthetic pigment, chlorophyll d, as the major reactive agent. The oxygenic photosynthesis based on this pigment may have evolved as an acclimatization to far-red light environments, or an as intermediate between the red-absorbing oxygenic and the far-red-absorbing anoxygenic photosynthesis that uses bacteriochlorophylls. Because of the unusual ratio of chlorophyll a to chlorophyll d in this organism, it has been used as a model to study the spectrographic characteristics of the two pigments. Acaryochloris marina MBIC11017. Acaryochloris marina MBIC11017 was isolated from algae from the coast of the Palau Islands in the western Pacific. This organism will be used for comparative analysis with other photosynthetic microorganisms. 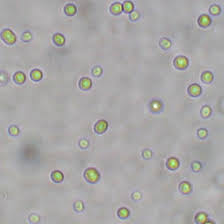
Lyngbya majuscula
en.wikipedia.org/wiki/Lyngbya_majuscula
Lyngbya majuscula is a species of cyanobacteria in the genus Lyngbya. Lyngbya majuscula grows on seagrass and is one of the causes of the human skin irritation seaweed dermatitis.It is known as fireweed in Australia and stinging limu in Hawai'.
The prevalence of this organism appears to be on the increase due to pollution and overfishing. Nutrients such as nitrogen and human waste flow to the ocean due to rain and sewage runoff; these added nutrients increase the population of microbes, which in turn remove oxygen from the water. Reduced numbers of fish to eat the microbes further enhances the microbe populations. Cyanobacteria are evolutionarily optimized for environmental conditions of low oxygen. Lyngbya majuscula can also fix its own nitrogen from atmospheric nitrogen dissolved in the seawater. This implies that pollution with phosphorus and iron may be more important than nitrogen pollution.
L. majuscula is known for its toxicity, producing "antifungal and cytotoxic agents, including laxaphycin A and B and curacin A."  Jeany |
|
|
|
Post by jeany on Jun 12, 2009 11:10:00 GMT -5
Aphanizomenonen.wikipedia.org/wiki/AphanizomenonAphanizomenon is an important genus of cyanobacteria that inhabits freshwater lakes and can cause choking blooms. Studies on the species Aphanizomenon flos-aquae have shown that it can regulate buoyancy through light-induced changes in turgor pressure.It is also able to move by means of gliding, though the specific mechanism by which this is possible is not yet known. 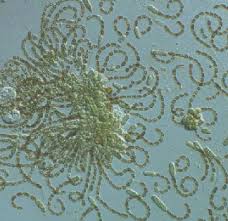 Anabaenaen.wikipedia.org/wiki/Anabaena Anabaenaen.wikipedia.org/wiki/AnabaenaAnabaena is a genus of filamentous cyanobacteria, or blue-green algae, found as plankton. It is known for its nitrogen fixing abilities, and they form symbiotic relationships with certain plants, such as the mosquito fern.They are one of four genera of cyanobacteria that produce neurotoxins, which are harmful to local wildlife, as well as farm animals and pets.
A DNA sequencing project was undertaken in 1999, which mapped the complete genome of Anabaena, which is 7.2 million base pairs long. The study focused on heterocysts, which convert nitrogen into ammonia. Certain species of Anabaena have been used on rice paddy fields, proving to be an effective natural fertilizer. 
Chroococcidiopsisnai.nasa.gov/news_stories/news_detail.cfm?ID=211What makes Chroococcidiopsis such a good candidate is its ability to survive in a wide range of extreme environments that are hostile to most other forms of life. Chroococcidiopsis has been found growing in hot springs, in hypersaline habitats, in a number of hot, arid deserts throughout the world, and in the frigid Ross Desert in Antarctica. "Chroococcidiopsis is the constantly appearing organism," Friedmann points out, "in nearly all extreme environments - at least extreme dry, extreme cold, and extremely salty environments. This is the one which always comes up." Moreover, where Chroococcidiopsis survives, it is often the only living thing that does. But it gladly gives up its dominance when conditions enable other, more complex forms of life to thrive. For clues on how to farm Chroococcidiopsis on Mars, Friedmann looks to its growth habits in arid regions on Earth. In desert environments, Chroococcidiopsis grows either inside porous rocks (endolithic growth), or just underground, on the lower surfaces of translucent pebbles (hypolithic growth). The pebbles provide an ideal microenvironment for Chroococcidiopsis in two ways. First, they trap moisture underneath them. Experiments have shown that small amounts of moisture can cling to the undersurfaces of rocks for weeks after their above-ground surfaces have dried out. Second, because the pebbles are translucent, they allow just enough light to reach the organisms to sustain growth.  
I think this last one is highy suspicious...just look at the pictures...and it can survive even under extreme circumstances...
I have also read the NASA has been doing tests on planet Mars with this bacterium...to test it's ability to grow and survive in atmospheric conditions.....without oxygen....www.techno-science.net/forum/viewtopic.php?t=204Jeany
|
|
|
|
Post by jeany on Jun 12, 2009 12:06:20 GMT -5
Nitrogen pollution harming ecosystems and contributing to global warming news.mongabay.com/2008/0515-nitrogen.htmlThe research, which involved dozens of scientists from around the world, shows that human activity is dramatically altering nitrogen cycles in Earth's oceans, soils, and atmosphere. The papers report that agricultural runoff and the burning of fossil fuels have boosted the supply of reactive nitrogen in the open oceans 50 percent above the normal range. The first paper, led by Robert Duce of Texas A&M University, found that nitrogen produced by human activity is responsible for about a third of the nitrous oxide and a tenth of the carbon dioxide input to the world's oceans each year. The researchers say the excess nitrogen "can deplete essential oxygen levels in the water and has significant effects on climate, food production, and ecosystems all over the world," according to a statement from Science. 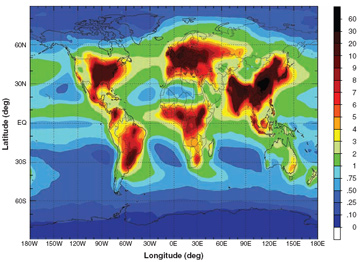 Estimated N deposition from global total N (NOy and NHx) emissions, totaling 105 Tg N per year. Courtesy of Science. Duce and colleagues calculate that humans account for up to three percent of the new marine biological production annually. While the increased biological activity sequesters CO2 from the atmosphere, the process produces nitrous oxide (N20), a greenhouse gas far more potent than carbon dioxide. "This fertilization of the ocean by human activities has an important impact on the exchange of the greenhouse gases carbon dioxide and nitrous oxide and should be considered in future climate change scenarios," Duce, a professor at Texas A&M University, said. "Anyone concerned about climate change will be alarmed at the scale of man's impact on the world's oceans, as revealed by our new study," added Peter Liss, a co-author of the paper and an environmental scientist at the University of East Anglia. "The natural nitrogen cycle has been very heavily influenced by human activity over the last century — perhaps even more so than the carbon cycle — and we expect the damaging effects to continue to grow. It is vital that policy makers take action now to arrest this. "The solution lies in controlling the use of nitrogen fertilizer and tackling pollution from the rapidly increasing numbers of cars, particularly in the developing world," he continued. Jeany |
|
|
|
Post by kammy on Jun 13, 2009 0:00:48 GMT -5
Synechococcus I believe this is a positive ID to a strain of cyanobacteria in Experiment 1. I have some microscopic shots of where it looks like someone has taken their finger and drawn in sand concentric circles just like in this photo below. I will see if I can locate my photo versions? "Synechococcus (from the Greek synechos (in succession) and the Greek kokkos (berry) ) is a unicellular cyanobacterium that is very widespread in the marine environment. Its size varies from 0.8 µm to 1.5 µm. The photosynthetic coccoid cells are preferentially found in well–lit surface waters where it can be very abundant (generally 1,000 to 200,000 cells per millilitre). Many freshwater species of Synechococcus have also been described. The genome of Synechococcus elongatus strain PCC7002 has a size of 2.7 Mbp, that of the oceanic strain WH8102 is 2.4 Mbp." *I believe I have photographed the elongated strain, also. If we could figure out its size - we could figure out if we are dealing with marine or fresh water. |
|
|
|
Post by kammy on Jun 13, 2009 0:02:11 GMT -5
Synechococcus IdentificationEar Lesion cultured in Agar @100x ![]() my-stuff-dot-com.com/My my-stuff-dot-com.com/My Stuff/Personal/Morgellons/PHOTO MASTERS/Experiment 2.1/3 18 09 Left Ear 2/03_19_2.JPEG [/img] ![]() my-stuff-dot-com.com/My my-stuff-dot-com.com/My Stuff/Personal/Morgellons/PHOTO MASTERS/Experiment 2.1/3 18 09 Left Ear 2/03_19_16.JPEG[/img] @450x ![]() my-stuff-dot-com.com/My my-stuff-dot-com.com/My Stuff/Personal/Morgellons/PHOTO MASTERS/Experiment 2.1/3 24 09 Left Ear 450x/03_24_69.JPEG[/img][/CENTER] |
|
|
|
Post by kammy on Jun 13, 2009 0:56:43 GMT -5
Cyanobacteria From Kat's research, this site shows some different types of cyanobacteria: CyanobacteriaWe determined that I possibly have cyanobacteria in my drinking water. I need to bring a couple of those photos over here, not as links but as images. Cyanobacteria is in the water from the factories in China that is producing some of our paper, cotton and cosmetics (and other products). Cyanobacteria contains a biofilm. |
|
|
|
Post by kammy on Jun 13, 2009 1:00:06 GMT -5
Cyanobacteria and the Nematode Connection Odor compounds from cyanobacterial biofilms acting as attractants and repellents for free-living nematodes" Document titleOdor compounds from cyanobacterial biofilms acting as attractants and repellents for free-living nematodes AbstractNematodes can both taste and smell an array of compounds, but whether and how these senses effect their capacity to locate microhabitats in aquatic environments is not known. Cyanobacterial biofilms may offer structure, shelter, and food for nematodes and are known to produce a variety of odor compounds. Our experiments demonstrate that aquatic nematodes are attracted to cyanobacterial biofilms using odor compounds as chemical cues. In contrast to the model organism, C. elegans, the chemotaxis of the aquatic nematode is elicited by a multicomponent odor rather than by single compounds." |
|
|
|
Post by kammy on Jun 13, 2009 1:19:45 GMT -5
My tap water 3 days growth, cultured in agar @100x: ![]() my-stuff-dot-com.com/My my-stuff-dot-com.com/My Stuff/Personal/Morgellons/PHOTO MASTERS/Water/5 14 09 5 11/05_14_11.JPEG [/img] ![]() my-stuff-dot-com.com/My my-stuff-dot-com.com/My Stuff/Personal/Morgellons/PHOTO MASTERS/Water/5 14 09 5 11/05_14_13.JPEG[/img] ![]() my-stuff-dot-com.com/My my-stuff-dot-com.com/My Stuff/Personal/Morgellons/PHOTO MASTERS/Water/5 14 09 5 11/05_14_14.JPEG[/img][/CENTER] |
|
|
|
Post by kammy on Jun 13, 2009 1:23:36 GMT -5
Calcification in cyanobacterial biofilms of alkaline salt lakes journals.cambridge.org/action/displayAbstract?fromPage=online&aid=47615"In contrast to biofilm-covered surfaces, calcium carbonate minerals nucleate and grow rapidly at surfaces poor in EPS when the critical supersaturation level for non-enzymatically controlled carbonate precipitation is reached. Examples of such surfaces poor in EPS are dead, lysed green algal cells and thin, discontinuous biofilms in voids of microbial reef rocks. Calcium carbonate crystals directly linked to cyanobacterial cells or filaments have been observed only exceptionally, e.g. on Calothrix." Calcium carbonate en.wikipedia.org/wiki/Calcium_carbonate"Calcium carbonate is a chemical compound with the chemical formula CaCO3. It is a common substance found in rock in all parts of the world, and is the main component of shells of marine organisms, snails, pearls, and eggshells." For those having parasitic snail/slug problems:"Weak acids such as acetic acid will react, albeit less vigorously. All of the rocks/minerals mentioned above will react with acid." DRINK APPLE CIDER VINEGAR! Start with a shot glass in the morning, one in the evening for 1 week. Then decrease to 1 shot glass per day until symptoms decrease. |
|


 lol... ok, like to kill like...
lol... ok, like to kill like...










